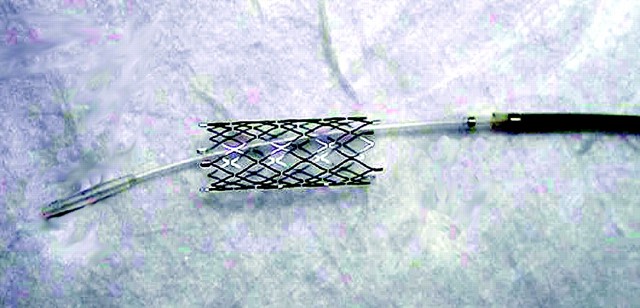
FDA Ignores Evidence Linking Wingspan Stent System to Stroke and Death, Keeps Dangerous Device on the Market
Statement of Dr. Sidney Wolfe, Director, Public Citizen’s Health Research Group
Note: The U.S. Food and Drug Administration today rejected Public Citizen’s Dec. 21, 2011 petition to ban the Wingspan Stent System.
In announcing the decision not to ban the Wingspan brain stent system, as Public Citizen had petitioned the agency to do last year, Dr. Jeff Shuren, the U.S. Food and Drug Administration’s (FDA) director of the Center for Device and Radiologic Health, said that “Patient benefit is an important factor in agency decision-making.”
Given the findings of the Stenting and Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis (SAMMPRIS) study, this statement misleadingly omits the fact that the patient benefits were clearly outweighed by the risks of the Wingspan Stent System in that study.
The SAMMPRIS was the only randomized controlled study on this device to compare the use of the Wingspan Stent System and aggressive medical management to aggressive medical management alone. Enrollment in the SAMMPRIS study was stopped because of a much higher 30-day rate of stroke or death in the Wingspan Stent group compared to the medical-management group (14.7 percent compared to 5.8 percent).
Thus, the evidence from this study showed that for every 11 patients treated with the Wingspan system and aggressive medical therapy, one additional patient dies or suffers a stroke within 30 days, compared to patients treated with aggressive medical therapy alone.
The FDA has largely ignored the findings of this important National Institutes of Health-funded study and has decided to allow this device to stay on the market where it is certain, despite labeling changes, to be used on patients similar to those in the SAMMPRIS study, because of the entirely predictable off-label uses that occur with such devices. Even for those patients getting the device according to the new labeling, it is not clear that the benefits outweigh the risks. This is further evidence that the agency is not making decisions in an evidence-based manner but, rather, stating that it believes the device will be beneficial, despite evidence to the contrary.
To view the original petition visit http://www.citizen.org/petition-to-fda-to-withdraw-approval-of-wingspan-stent-system-122111